A
cross-sectional
study
was
done
on
asthmatics
whose
diagnosis
had
been
clinically
confirmed
by
physical
examination
and
pulmonary
function
test
(PFT)among
pregnant
women
who
were
referred
to
Mobini
Hospital
in
Sabzevar
between
August
2014
and
April
2015.
The
Ethics
Committee
of
Sabzevar
University
approved
the
study
protocol.
The
Radio-allergo-sorbent
test
(RAST)
was
done,
using
allergen
extracts,
to
determine
the
patients'
sensitivity
to
food
and
inhalation
allergens.
(20).
RASTs
were
carried
out
using
standardized
allergen
extracts
for
the
following
36
aeroallergens
and
36
food
allergen:
aspergillus
fumigatus,
cat,
dog,
cow,
sheep,
cage
bird
mix,
sweet
vernal
grass,
horse,
Alternaria
alternata,
Cladosporium
herbarum,
pollens
[tree
(Ashe,
Tree
mixture),
(Russian
thistle,],
mites
(Dermatophagoides
pteronyssinus,
Dermatophagoides
farine),
feathers
mixture,
cockroach
peanut,
seafood
mix
3,
banana,
orchard
grass,
cultivated
rye,
alder,
penicillium
notatum,
English
plantain,
goosefoot,
rough
pigweed,
rice,
grain
mix
2,
apple,
cultivated
oat,
meadow
foxtail,
firebush,
sorrel,
tree
mix
6,
honeybee
venom
1,
house
dust,
cross-reactive
carbohydrate
determinants
(CCD)
marker,
shrimp
prawn,
white
pine,
tree
mix
4,
rye
flour,
potato,
rose,
cultivated
oat.
From
those
mothers
with
clinically-confirmed
asthma,
in
the
final
weeks
of
pregnancy,
7
cc
of
blood
was
drawn
for
total
IgE
and
RAST.
Additional
blood
samples
from
their
neonates
were
obtained
first
day
after
delivery
for
the
identical
lab
tests.
The
clot
sample
for
allergens
samples
were
transported
to
the
laboratory
quickly.
Total
serum
was
kept
at
-20°
C
until
testing.
Allergen
samples
for
each
series
of
experiments
were
stored
at
4
°
C
for
up
to
2
weeks,
collected
and
tested.
This
methodology
continued
until
all
the
required
samples
were
gathered.
Tests
were
performed
using
the
ELISA
kit
(Euroimmun,
Germany).
In
those
patients
with
allergies
to
a
wide
variety
of
allergens,
additional
inhalation
and
food
allergens
were
tested.
Blood
samples
were
processed
in
accordance
with
kit
instructions,
i.e.
based
upon
the
multiplex
immunoblots
method.
Then
the
results
of
total
serum
IgE,
using
standard
and
allergens
results,
were
evaluated
with
the
software.
Mothers
were
considered
to
be
atopic
if
they
had
IgE
?160
kIU/L,
infants
if
their
serum
IgE
?10
kIU/L.
Any
additional
patient
history
was
also
recorded.
The
results
were
analysed
using
SPSS
version
20.
Cohort
description
Out
of
1603
pregnant
women
34
(2.1%)
had
asthma.
The
majority
of
asthmatic
patients
were
either
25-29
years
(29
%)
or
?35
years
(35.3%)
of
age.
The
frequency
of
atopy
in
the
study
population
of
34
asthmatic
mothers
and
their
infants
was
calculated
as
7
(21%)
and
4
(12%),
respectively.
Some
allergens
were
much
more
common
than
others.
(Tables
1,
2).
Food
allergy
was
defined
as
sensitization
to
at
least
1
food
allergen
and
was
present
in
28%.
In
a
multivariate
analysis,
we
found
a
significant
relationship
between
prematurity
and
atopy
or
asthma
(P
=
0.006).
Allergies
of
pregnant
women
are
related
to
asthma
To
describe
potential
factors
that
might
be
connected
to
asthma
during
pregnancy
(AP),
we
included
the
following
parameters:
eczema,
allergies,
asthma
severity,
asthma
control,
atopy,
IgE,
seafood
mix
3
and
rough
pigweed.
As
controls,
the
same
parameters
were
tested
in
the
non
-asthmatic
mothers.
The
data
were
analysed
using
the
chi-square
statistic
and
Fischer's
exact
test.
The
highest
rates
of
allergies
were
in
the
asthmatic
group:
32
(94.1%)
(p
=
0.001).
The
majority
of
subjects
in
the
asthmatic
group
did
not
have
eczema,
30
(88.2
%).
Asthmatic
patients
were
divided
into
those
with
intermittent/mild
persistent
asthma
(IMPA),
and
moderate
or
severe
persistent
asthma
(MSPA).
We
found
no
significant
relationship
between
atopy
and
asthma
severity,
with
22%
and
14%
atopy
in
IMPA
and
MSPA
respectively.
As
well,
there
was
no
significant
relationship
between
asthma
control
(i.e
well-controlled,
vs.
partly/poorly-controlled)
and
atopy
with
23%
and
19%
atopy,
respectively.
There
was
no
association
between
atopy,
and
allergy
or
eczema
(Fischer's
exact
test
p=0.37
and
p=1.0,
respectively).
There
was
also
no
significant
statistical
association
between
atopy
and
seafood
mix
3
or
rough
pigweed,
(both
p=1.0)
or
any
other
allergen
tested.
Hence,
it
appeared
that
only
having
allergy
at
pregnancy
was
closely
linked
to
asthma,
and
it
would
be
interesting
to
determine
whether
asthma
might
also
be
linked
to
additional
health
issues.
Atopy
and
location-dependent
wheezing
or
exercise-induced
wheezing
of
pregnant
women
are
related.
To
investigate
whether
further
factors
might
also
have
contributed
to
AP,
we
tested
the
following
factors:
asthma
severity,
common
allergens,
smoker,
passive
smoker,
atopy
and
infant
gender.
There
was
a
positive
statistical
association
between
atopy
and
location-dependent
wheezing
or
exercise-induced
wheezing,
p=0.048
and
p=0.004,
respectively.
There
were
no
association
between
asthma
severity
and
the
most
common
allergen
in
asthmatic
mothers,
p=0.71
(Table
3).
Table
4
shows
the
distribution
of
the
variables
retrieved
from
the
databases
for
atopic
women
included
in
the
study.
There
was
no
significant
relationship
between
atopy
and
smoking
or
passive
smoking
p=0.51
and
p=0.58,
respectively.
Taken
together,
it
appeared
that
both
allergy
and
atopy
and
location-dependent
wheezing
and
exercise-induced
wheezing
in
pregnant
women
were
associated
with
a
higher
prevalence
of
asthma.
Table
1:
Positive
RAST
results
in
cord
and
infant
blood
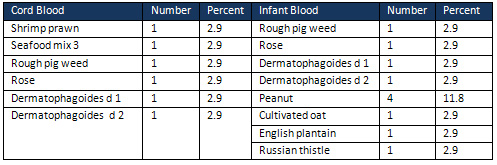
Table
2:
Positive
RAST
results
in
pregnant
women
with
asthma
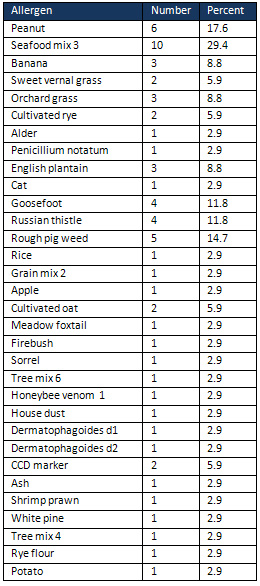 Table
3:
Frequency
of
positivity
to
seafood
mix
3
in
relation
to
asthma
severity
Table
3:
Frequency
of
positivity
to
seafood
mix
3
in
relation
to
asthma
severity
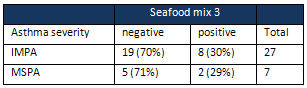
IMPA:
intermittent
asthma/mild
persistent
asthma;
MSPA…moderate
and
severe
persistent
asthma
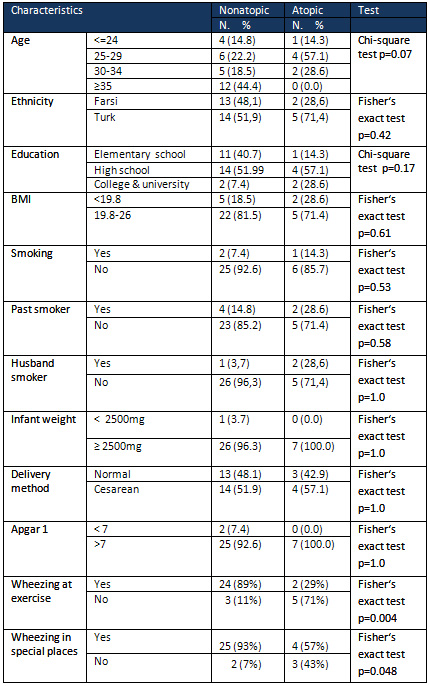
Table
4:
Characteristics
of
pregnant
women
with
asthma
by
atopy
status
This
study
is
the
first
in
Sabzevar
to
assess
the
prevalence
of
asthma
and
allergic
sensitization
and
its
association
with
IgE
factors
in
pregnant
women
with
asthma.
Sabzevar
is
a
city
in
north-eastern
Iran.
The
region
is
semi
desert
with
hot
and
dry
summers
and
is
an
agricultural
center
for
grape
and
raisin
farming.
In
the
present
study,
we
looked
into
the
possible
connection
between
asthma
and
allergens.
We
found
a
significant
relationship
between
allergies
and
asthma
as
well
as
atopy
and
location-related
and
exercise-induced
wheezing
in
pregnant
women.
Atopy
and
asthma
severity,
atopy
and
allergic
sensitisations
to
various
food
and
inhalation
allergens
were
not
statistically
significantly
related.
We
observed
that
food
allergens
were
more
common
than
aeroallergens
in
both
mothers
and
infants.
The
frequency
of
atopy
in
asthmatic
mothers
and
their
infants
were
21%
and
12%,
respectively.
Our
results
are
similar
to
those
of
Nabavi
(2013).
(14)
It
is
possible
that
there
are
relations
between
the
in
utero
environment
in
asthmatic
pregnancies
and
the
development
of
asthma
during
childhood,
independent
of
genetic
factors.
This
is
suggested
by
atopy
in
children,
which
was
more
frequently
related
to
maternal
asthma
or
IgE
levels,
rather
than
paternal
asthma
or
IgE.(15)
The
study
prospectively
followed
181
AP,
62%
of
whom
were
classified
as
atopic.(16)
Comert
reported
a
prevalence
of
32.2%
atopic
cases
in
his
study
(17).
This
difference
as
compared
with
the
findings
in
the
present
study
is
probably
due
to
climatic
factors
(12).
This
study
is
the
first
report
of
sensitization
to
food
allergens
in
our
region.
A
high
rate
of
sensitisation
to
pollens
was
established
in
earlier
studies
in
our
country.
A
study
in
Mashhad
(18),
Iran
reported
that
weeds
had
the
highest
rate
of
sensitization,
among
which
Russian
thistle
(salsolakali)
was
the
most
common
in
childhood.
This
result
is
similar
to
our
results
in
infants
but,
probably
due
to
the
dry
climate
or
different
method
of
testing,
it
was
not
the
most
common.
Whereas
we
used
the
RAST,
they
applied
the
skin
prick
test.
Also,
in
Shiraz,
the
prevalence
pattern
of
sensitisations
to
different
types
of
pollens
(e.g
weed,
grass,
trees),
as
well
as
in
Tehranand
Karaj
(12)
(herbacee
I/II/III,
sycamore,
chenopodium,
tree
mix,
grass,
ash
and
cedar)
was
similar
to
our
study.
In
the
Comert
(2014)
study,
the
most
prevalent
allergen
was
Phleumpratense
(19.3%),
whereas
in
our
study,
it
was
seafood
mix
3
(29.4%).
(17)
In
our
study,
food
allergens
were
the
most
common
in
mothers
and
grass
and
peanut
allergens
in
infants.
Herbal
topography,
climate
and
temperature
could
be
responsible
for
the
difference.
(18)
Our
results
were
similar
to
most
European
countries
in
that
three
to
four
allergens
were
recognized
in
95%
of
all
sensitized
subjects.
(17)
It
is
well-known
that
allergic
patients
are
commonly
co-sensitized
against
different
allergen
sources.
Advancement
in
the
field
of
allergen
description
by
molecular
biological
techniques
has
now
shown
that
sensitization
against
different
allergen
sources
can
be
clarified
as
cross-reactivity
of
IgE
antibodies
with
fundamentally
and
immunologically
linked
components
existent
on
these
allergen
sources.
(12)
A
survey
in
Kerman
indicated
that
allergies
to
food
and
airborne
allergens
differ
depending
on
the
nutritional
and
environmental
settings.
(19)
The
sensitization
rates
to
grass
pollen
varies
between
19.5%
and
69.9%
among
European
countries,
and
it
is
the
most
common
allergen
in
Austria,
Denmark,
England,
Greece,
Poland,
Switzerland
and
the
Netherlands
(17).
In
contrast
in
Comert's
study,
the
prevalence
of
Dermatophagoides
pteronyssinus
and
Dermatophagoides
farinae
was
also
high
16.2%
and
10.5%,
respectively.
The
sensitization
rates
to
Dermatophagoides
pteronyssinus
varied
between
16.8%
and
68.8%
within
European
countries.
Indeed,
it
is
the
most
common
allergen
in
Belgium,
France,
Italy
and
Portugal.
In
our
results,
the
frequency
was
2.9%.
Two
studies
in
Turkey
reported
the
prevalence
of
sensitization
to
allergens
from
cats
and
dogs.
There
is
a
growing
tendency
towards
pet
ownership
in
our
country
(20,
21),
so
this
may
become
a
problem
in
the
future.
In
Comert's
study,
the
sensitization
rates
to
Alternaria,
Aspergillus
and
Cladosporium
were
1.1%,
0.9%
and
0.7%,
respectively.
In
Europe
(22),
however,
the
rate
of
Cladosporium
allergies
was
reported
to
be
between
0%
and
12.8%.
The
cockroach
allergen
would
have
been
an
important
allergen
to
assess
if
we
had
been
able
to
test
all
of
the
subjects
against
this
allergen.
Mert's
study
reported
74.22%
cockroach
sensitisations
(17)
Whereas
a
study
in
Ahvaz
found
the
most
prevalent
sensitizing
mould
was
C.
acremonium
followed
by
Penicillium
spp,
we
did
not
find
that
the
infants
were
sensitized
to
these;
although
we
tested
them
for
Penicillium
spp
and
Aspergillus
allergen.
(24)
Penicillium,
aspergillus,
and
basidiospores
are
some
of
the
fungal
species
that
are
associated
with
asthma
and
atopy
in
children
(25).
We
found
only
one
such
subject.
Bunyavanich
(2014)
reported
that
exposure
to
wheat
during
early
pregnancy
was
associated
with
reduced
odds
of
mid-childhood
allergy
and
asthma.
Alterations
in
immune
function
have
been
proposed
as
a
mechanism
contributing
to
later
development
respiratory
disease
(26).
Since
we
have
only
investigated
neonates,
the
further
development
of
this
group
is
a
matter
of
further
study.
The
group
of
34
asthmatic
patients
tested
with
the
36
food
and
36
aeroallergen
panel
were
comparatively
young
which
could
overestimate
the
prevalence
of
allergen
sensitivity.(17)
Our
findings
suggest
a
negative
relationship
between
location
and
exercise-related
wheezing
and
atopy
and
a
slightly
positive
association
between
atopy
and
maternal
and
passing
smoking.
We
found
no
statistically
significant
associations
between
atopy
and
method
of
delivery,
parity,
number
of
gravidities,
and
abortions.
However,
Pistiner's
(2008)
findings
suggest
that
cesarean
delivery
is
associated
with
allergic
rhinitis
and
atopy
among
children
with
a
parental
history
of
asthma
or
allergies.
This
likely
differs
from
our
study
due
to
the
alternative
characteristics
of
the
populations
we
each
studied,
as
well
as
the
duration
of
follow-up
and
definition
of
the
outcomes.
(23)
The
Mean
(SD)
age
of
our
asthmatic
patient
group
30.97
±6.36
versus
27.33
±5.91
seems
to
be
lower
than
in
the
Spanish
study,
i.e,
36.2
±12.72
versus
30±15
years,
respectively,
which
could
account
for
the
lower
prevalence
of
atopic
sensitization
found
in
our
study.
Another
possibility
may
be
that
the
Spanish
study
consisted
mainly
of
patients
with
allergic
rhinitis;
with
37%
of
them
having
asthma.
(17)
Our
main
limitation
was
the
lack
of
lab
tests
in
the
control
group.
A
smaller
sample
size
in
contrast
to
earlier
studies
was
another
weakness
of
our
study.
According
to
the
results
and
environmental
condition
in
the
area
of
our
study,
the
most
common
regional
allergens
were
food
allergens
such
as
seafood
mix
3
and
peanut
that
should
be
given
more
attention.
It
may
be
important
in
future
studies
to
try
to
detect
local
allergens
that
have
a
potential
of
materno-fetal
transfer.
Acknowledgments
This
work
was
approved
by
the
ethics
committee
of
the
Medical
University
of
Sabzevar,
Iran
(Medsab
Rec.93.36).
We
thank
the
vice-chancellor
of
the
Sabzevar
university
-Iran
for
financial
support
of
this
research
.We
thank
the
personnel
of
Mobini
and
Vaseei
Hospitals
for
their
cooperation.
We
also
thank
all
the
mothers
who
participated
in
the
study.
1.
Ozol
D,
Mete
E.
Asthma
and
food
allergy.
Current
opinion
in
pulmonary
medicine.
2008;14(1):9-12.
2.
de
Silva
NR,
Dasanayake
WMDK,
Karunatilleke
C,
Malavige
GN.
Food
dependant
exercise
induced
anaphylaxis
a
retrospective
study
from
2
allergy
clinics
in
Colombo,
Sri
Lanka.
Allergy,
Asthma
&
Clinical
Immunology.
2015;11(1):1.
3.
Pedrosa
M,
Boyano-Martínez
T,
García-Ara
C,
Caballero
T,
Quirce
S.
Utility
of
specific
IgE
to
Ara
h
6
in
peanut
allergy
diagnosis.
Annals
of
Allergy,
Asthma
&
Immunology.
2015;115
(2):108-12.
4.
Pedrosa
M,
Boyano-Martínez
T,
García-Ara
MC,
Caballero
T,
Quirce
S.
Peanut
seed
storage
proteins
are
responsible
for
clinical
reactivity
in
Spanish
peanut?allergic
children.
Pediatric
Allergy
and
Immunology.
2012;23(7):654-659.
5.
Ackerbauer
D,
Bublin
M,
Radauer
C,
et
al.
Component-resolved
IgE
profiles
in
Austrian
patients
with
a
convincing
history
of
peanut
allergy.
International
archives
of
allergy
and
immunology.
2015;166(1):13-24.
6.
Zheng
X-y,
Ding
H,
Jiang
L-n,
et
al.
Association
between
air
pollutants
and
asthma
emergency
room
visits
and
hospital
admissions
in
time
series
studies:
a
systematic
review
and
meta-analysis.
PloS
one.
2015;10(9):e0138146.
7.
Bunyavanich
S,
Rifas-Shiman
SL,
Platts-Mills
TA,
et
al.
Peanut,
milk,
and
wheat
intake
during
pregnancy
is
associated
with
reduced
allergy
and
asthma
in
children.
Journal
of
Allergy
and
Clinical
Immunology.
2014;133(5):1373-1382.
8.
Saadeh
D,
Salameh
P,
Caillaud
D,
et
al.
Prevalence
and
association
of
asthma
and
allergic
sensitization
with
dietary
factors
in
schoolchildren:
data
from
the
french
six
cities
study.
BMC
public
health.
2015;15(1):1.
9.
Schwela
D.
Air
pollution
and
health
in
urban
areas.
Reviews
on
environmental
health.
2000;
15(1-2):13-42.
10.
de
Blay
F,
Doyen
V,
Lutz
C,
et
al.
A
new,
faster,
and
safe
nasal
provocation
test
method
for
diagnosing
mite
allergic
rhinitis.
Annals
of
Allergy,
Asthma
&
Immunology.
2015;115(5):385-390.
e381.
11.
Kim
H-J,
Kang
M-H,
Park
H-M.
Common
allergens
of
atopic
dermatitis
in
dogs:
comparative
findings
based
on
intradermal
tests.
Journal
of
veterinary
science.
2011;12(3):287-290.
12.
Singh
AB,
Mathur
C.
An
aerobiological
perspective
in
allergy
and
asthma.
Asia
Pacific
Allergy.
2012;2(3):210-222.
13.
Zicari
A,
Indinnimeo
L,
De
Castro
G,
et
al.
Food
allergy
and
the
development
of
asthma
symptoms.
International
journal
of
immunopathology
and
pharmacology.
2012;25(3):731-740.
14.
Nabavi
M,
Ghorbani
R,
Asadi
AM,
Faranoush
M.
Factors
associated
with
cord
blood
IgE
levels.
Asian
Pacific
Journal
of
Allergy
and
Immunology.
2013;31(2):157.
15.
Murphy
V,
Gibson
P,
Smith
R,
Clifton
V.
Asthma
during
pregnancy:
mechanisms
and
treatment
implications.
European
Respiratory
Journal.
2005;25(4):731-750.
16.
Stenius-Aarniala
B,
Piirilä
P,
Teramo
K.
Asthma
and
pregnancy:
a
prospective
study
of
198
pregnancies.
Thorax.
1988;43(1):12-18.
17.
Comert
S,
Demir
AU,
Karakaya
G,
Kalyoncu
AF.
Minimum
prick
test
panel
for
adult
patients
with
asthma
and
rhinitis
in
Ankara,
Turkey.
Journal
of
Asthma.
2014;51(4):417-422.
18.
Behmanesh
F,
Shoja
M,
Khajedaluee
M.
Prevalence
of
aeroallergens
in
childhood
asthma
in
Mashhad.
Macedonian
Journal
of
Medical
Sciences.
2010;3(3):295-298.
19.
Fouladseresht
H,
Safiri
S,
Moqaddasi
M,
Razeghi
MS,
Bazargan
N.
Prevalence
of
food
and
airborne
allergens
in
allergic
patients
in
Kerman.
Journal
of
Kermanshah
University
of
Medical
Sciences
(J
Kermanshah
Univ
Med
Sci).
2014;18(4):234-241.
20.
Bostanci
I,
Türktas
I,
Türkyilmaz
C.
Sensitization
to
aeroallergens
in
Ankara,
Turkey.
Allergy.
1999;54:1332-1334.
21.
Füsun
KA.
Cockroach
sensitivity
in
inner-city
allergic
patients
in
Turkey.
Turkish
Respiratory
Journal.
2001;2(1):17-20.
22.
Heinzerling
L,
Burbach
G,
Edenharter
G,
et
al.
GA2LEN
skin
test
study
I:
GA²LEN
harmonization
of
skin
prick
testing:
novel
sensitization
patterns
for
inhalant
allergens
in
Europe.
Allergy.
2009;64(10):1498-1506.
23.
Pistiner
M,
Gold
DR,
Abdulkerim
H,
Hoffman
E,
Celedón
JC.
Birth
by
cesarean
section,
allergic
rhinitis,
and
allergic
sensitization
among
children
with
a
parental
history
of
atopy.
Journal
of
Allergy
and
Clinical
Immunology.
2008;122(2):274-279.
24.
Yang
H,
Xun
P,
He
K.
Fish
and
fish
oil
intake
in
relation
to
risk
of
asthma:
a
systematic
review
and
meta-analysis.
PloS
one.
2013;8(11):e80048.
25.
Wen
HJ,
Chiang
TL,
Lin
SJ,
Guo
YL.
Predicting
risk
for
childhood
asthma
by
pre?pregnancy,
perinatal,
and
postnatal
factors.
Pediatric
Allergy
and
Immunology.
2015;26(3):272-279.
26.
Hollams
EM,
De
Klerk
NH,
Holt
PG,
Sly
PD.
Persistent
effects
of
maternal
smoking
during
pregnancy
on
lung
function
and
asthma
in
adolescents.
American
journal
of
respiratory
and
critical
care
medicine.
2014;189(4):401-407.

