The
patients
were
aged
21-60
years
who
were
76%
women
and
the
rest
=
men,
with
a
mean
age
of
34.8
years
for
women
and
37
years
for
men.
Mean
serum
level
of
Ca2+
was
3.5
mmol/L
and
4.5
mmol/L
in
women
during
the
headache
attacks
and
normal
respectively
and
Mean
serum
level
of
ionized
Ca2+
was
3.7
mmol/L
and
4.3
mmol/L
in
men
during
the
headache
attacks
and
normal
condition
(with
no
headaches)
respectively.
There
was
a
significant
difference
on
serum
Ca2+
level
during
migraine
attacks
decreased
significantly
compared
with
normal
mode
in
women
and
men
(P
values
<0.05).
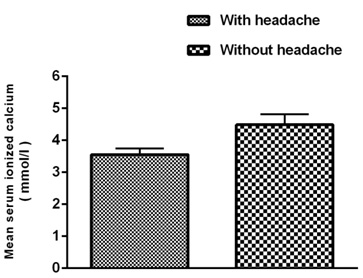
Figure
1:
Decreasing
ionized
calcium
level
during
migraine
headache
Figure
2:
Mean
serum
ionized
calcium
level
reduced
significantly
during
migraine
headache
in
women

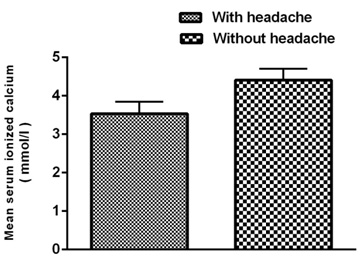
Figure
3:
Mean
serum
ionized
calcium
level
decreased
significantly
during
migraine
headaches
in
men
Migraine
seems
to
have
a
complex
pathogenesis
(2).
Several
theories
have
been
suggested
to
explain
the
mechanism
of
migraine.
A
phosphorylation
oxidative
defect,
malfunction
of
intra
neuronal
voltage
gated
calcium
channels,
intracellular
magnesium
(Mg)
deficiency
or
a
combination
of
these
may
make
the
cells
susceptible
to
spontaneous
depolarization
(17).
Many
patients
with
familial
hemiplegic
migraine
have
a
missense
mutation
in
the
P/Q
calcium
channel,
so
that
this
form
of
migraine,
at
least,
is
associated
with
a
demonstrable
calcium
channelopathy.
In
menstrual
migraine,
ionized
Mg
level
is
decreased
and
the
ratio
of
Ca/Mg
level
is
increased.
Therefore,
serum
Mg
level
may
have
a
role
in
pathogenesis
of
menstrual
migraine
(18).
The
results
of
the
present
study
showed
serum
ionized
calcium
level
decreased
substantially
in
migraine
headaches
compared
to
normal
subjects.
Also
examining
the
ionized
calcium
level
in
men
and
women
separately
showed
that
the
ion
level
in
both
genders
reduced
significantly
in
migraine
headaches
than
normal
subjects.
Findings
may
also
add
to
the
growing
evidence
for
involvement
of
magnesium
in
migraine
pathophysiology
(1).
Other
studies
show
that
Ca2+
and
other
ion
channels
are
important
in
the
mechanism
of
cortical
spreading
depression,
which
is
believed
to
initiate
migraine
attacks
(2).
Thus,
impaired
function
of
cerebral
Ca2+
channels
may
facilitate
the
initiation
of
attacks
(10).
Recent
studies
have
also
concluded
that
abnormalities
in
the
channels
within
the
cells
that
transport
calcium,
magnesium,
sodium
and
potassium
contribute
to
the
onset
of
migraines
(3).
The
calcium
channels
are
known
to
regulate
the
release
of
serotonin.
with
impaired
functioning
of
the
channels,
serotonin
may
not
effectively
be
released
or
may
be
released
in
low
quantities
(2).
As
a
consequence
of
this
dip,
a
range
of
migraine
symptoms
begin
to
occur
(19).
Previous
study
suggests
that
the
abnormality
of
glucose
level,
insulin
resistance,
and
?-cell
function
have
significant
correlation
with
alteration
of
serum
calcium
homeostasis
(20).
As
these
factors
were
not
adjusted
in
our
study,
this
is
a
limitation
for
the
present
study.
Based
on
the
results
obtained
in
the
present
study,
there
was
a
significantly
decreased
serum
ionized
calcium
level
in
patients
during
migraine
attack
in
males
and
females
compared
to
in
normal
subjects
(those
with
no
headaches).
Acknowledgement
Author
thank
the
patients
who
that
participated
in
present
study.
Also,
the
present
study
has
been
supported
by
Deputy
of
research,
Jahrom
University
of
Medical
Sciences.
1.
Taga
A,
Russo
M,
Manzoni
GC,
Torelli
P.
Cluster
Headache
With
Accompanying
Migraine-Like
Features:
A
Possible
Clinical
Phenotype.
Headache.
2016.
2.
Verrotti
A,
Iapadre
G,
Pisano
S,
Coppola
G.
Ketogenic
diet
and
childhood
neurological
disorders
other
than
epilepsy:
an
overview.
Expert
Rev
Neurother.
2016.
3.
Ashrafi
MR,
Najafi
Z,
Shafiei
M,
Heidari
K,
Togha
M.
Cinnarizine
versus
Topiramate
in
Prophylaxis
of
Migraines
among
Children
and
Adolescents:
A
Randomized,
Double-Blind
Clinical
Trial.
Iran
J
Child
Neurol.
2014;8(4):18-27.
4.
Rabiee
B,
Zeinoddini
A,
Kordi
R,
Yunesian
M,
Mohammadinejad
P,
Mansournia
MA.
The
Epidemiology
of
Migraine
Headache
in
General
Population
of
Tehran,
Iran.
Neuroepidemiology.
2016;46(1):9-13.
5.
Thakur
KT,
Albanese
E,
Giannakopoulos
P,
Jette
N,
Linde
M,
Prince
MJ,
et
al.
Neurological
Disorders.
In:
Patel
V,
Chisholm
D,
Dua
T,
Laxminarayan
R,
Medina-Mora
ME,
editors.
Mental,
Neurological,
and
Substance
Use
Disorders:
Disease
Control
Priorities,
Third
Edition
(Volume
4).
Washington
(DC):
The
International
Bank
for
Reconstruction
and
Development
/
The
World
Bank
(c)
2016
International
Bank
for
Reconstruction
and
Development
/
The
World
Bank.;
2016.
6.
Reddy
KS.
Global
Burden
of
Disease
Study
2015
provides
GPS
for
global
health
2030.
The
Lancet.
2016;388(10053):1448-9.
7.
Tok
F,
Balaban
B,
Ya?ar
E,
Safaz
?,
Y?lmaz
B,
Alaca
R.
Ionized
calcium
levels
in
stroke
patients
and
its'
relation
with
hemiplegic
upper
limb
pain.
Age
(year).
2015;54:14.21.
8.
Kubicka-Baczyk
K,
Labuz-Roszak
B,
Pierzchala
K,
Adamczyk-Sowa
M,
Machowska-Majchrzak
A.
Calcium-phosphate
metabolism
in
patients
with
multiple
sclerosis.
Journal
of
endocrinological
investigation.
2015;38(6):635-42.
9.
Al-Hamzawi
AO,
Rosellini
AJ,
Lindberg
M,
Petukhova
M,
Kessler
RC,
Bruffaerts
R.
The
role
of
common
mental
and
physical
disorders
in
days
out
of
role
in
the
Iraqi
general
population:
results
from
the
WHO
World
Mental
Health
Surveys.
J
Psychiatr
Res.
2014;53:23-9.
10.
Mazaheri
S,
Poorolajal
J,
Hosseinzadeh
A,
Fazlian
MM.
Effect
of
intravenous
sodium
valproate
vs
dexamethasone
on
acute
migraine
headache:
a
double
blind
randomized
clinical
trial.
PLoS
One.
2015;10(3):e0120229.
11.
Slavin
M,
Bourguignon
J,
Jackson
K,
Orciga
MA.
Impact
of
Food
Components
on
in
vitro
Calcitonin
Gene-Related
Peptide
Secretion-A
Potential
Mechanism
for
Dietary
Influence
on
Migraine.
Nutrients.
2016;8(7).
12.
Malone
CD,
Bhowmick
A,
Wachholtz
AB.
Migraine:
treatments,
comorbidities,
and
quality
of
life,
in
the
USA.
2015.
13.
Siva
A,
Lampl
C.
Case-Based
Diagnosis
and
Management
of
Headache
Disorders:
Springer;
2015.
14.
Sava
L,
Pillai
S,
More
U,
Sontakke
A.
Serum
calcium
measurement:
total
versus
free
(ionized)
calcium.
Indian
Journal
of
Clinical
Biochemistry.
2005;20(2):158-61.
15.
Bowers
G,
Brassard
C,
Sena
SF.
Measurement
of
ionized
calcium
in
serum
with
ion-selective
electrodes:
a
mature
technology
that
can
meet
the
daily
service
needs.
Clinical
chemistry.
1986;32(8):1437-47.
16.
Fabry
P,
Fouletier
J.
Chemical
and
Biological
Microsensors:
Applications
in
Fluid
Media:
John
Wiley
&
Sons;
2013.
17.
Ansari
B,
Basiri
K,
Meamar
R,
Chitsaz
A,
Nematollahi
S.
Association
of
Helicobacter
pylori
antibodies
and
severity
of
migraine
attack.
Iran
J
Neurol.
2015;14(3):125-9.
18.
Mauskop
A,
Altura
BT,
Altura
BM.
Serum
ionized
magnesium
levels
and
serum
ionized
calcium/ionized
magnesium
ratios
in
women
with
menstrual
migraine.
Headache:
The
Journal
of
Head
and
Face
Pain.
2002;42(4):242-8.
19.
Liu
L,
Pei
P,
Zhao
LP,
Qu
ZY,
Zhu
YP,
Wang
LP.
Electroacupuncture
Pretreatment
at
GB20
Exerts
Antinociceptive
Effects
via
Peripheral
and
Central
Serotonin
Mechanism
in
Conscious
Migraine
Rats.
Evid
Based
Complement
Alternat
Med.
2016;2016:1846296.
20.
Sun
G,
Vasdev
S,
Martin
GR,
Gadag
V,
Zhang
H.
Altered
calcium
homeostasis
is
correlated
with
abnormalities
of
fasting
serum
glucose,
insulin
resistance,
and
?-cell
function
in
the
Newfoundland
population.
Diabetes.
2005;54(11):3336-9.

