|
Evaluation of seizures
in pregnant women in Kerman – Iran
Hossein Ali Ebrahimi (1)
Elahe Arabpour (2)
Kaveh Shafeie (3)
Narges Khanjani (4)
(1) M.D Professor of Neurology, Neurology Research
Center, Kerman University of Medical Sciences
Kerman, Iran
(2) M.D Assistant Professor of Neurology, Neurology
Department, Afzalipor Faculty, Kerman University
of Medical Sciences, Kerman, Iran
(3) M.D Assistant Professor of Neurology, Neurology
Research Center, Kerman University of Medical
Sciences Kerman, Iran
(4) M.D Associate Professor of Epidemiology,
Neurology Research Center, Kerman University
of Medical Sciences Kerman, Iran
Correspondence:
Hossein Ali Ebrahimi
M.D Professor of Neurology,
Neurology Research Center,
Kerman University of Medical Sciences
Kerman,
Iran
Email: hebrahimi@kmu.ac.ir
|
Abstract
Background and Objectives:
Seizure occurs in 0.5 to 1 percent of
pregnant women, marking it as one of the
most prevalent serious neurological disorders
during pregnancy. Women with epilepsy
face a greater threat of pregnancy-related
adverse effects. The effects of seizures
on pregnancy and its maternal and fetal
adverse effects, necessitates the study
of the prevalence of pregnancy seizures,
an issue ignored by previous studies conducted
elsewhere.
Method: The
present study is a descriptive-analytical
research. The participants included women
who were referred to delivery centers
of Kerman (public and private) for childbirth.
The exclusion criteria were lack of cooperation
and consent of the subjects for sitting
the interviews.
Results:
Among 3,807 admitted pregnant women, 38
cases (1%) experienced epileptic seizures.
Among the participants, 2,125 subjects
were admitted to public hospitals and
1,682 subjects to private hospitals. Seizure
recurrence remained constant in 58% of
the subjects, declined in 21%, and increased
in 21%. Twenty four cases (0.63%) had
a history of prepartum seizures, and 14
cases (0.36%) experienced seizures for
the first time during pregnancy, with
eclampsia as the most prevalent cause.
Patients of public hospitals had lower
levels of education and higher number
of epileptic seizures. Average age of
patients experiencing epileptic seizures
was lower than the non-epileptic cases.
Seizures were observed more in nulliparous
women.
Conclusion:
More than 6 in 1,000 pregnant women suffer
from epilepsy. Eclampsia is the most prevalent
cause. Epileptic seizures increased in
21% of epileptic pregnant women, and declined
in 21% of the cases.
Key words:
seizure, epilepsy, pregnant, women
|
Seizures are referred to as the temporary physiological
dysfunction of the brain, caused by the abnormal
electrical and excessive discharges of cortical
neurons, and epilepsy is the unexplained and
unpredicted repetition of these attacks (1-5).
Women with epilepsy have a greater risk of pregnancy-related
adverse effects, including cesarean section,
preeclampsia, pregnancy-induced hypertension,
premature contraction or preterm labor, postpartum
hemorrhage, possibility of stillbirth, and microcephaly.
Also, the risks of mental retardation and afebrile
seizures are increased in their infants. Women
with a history of pregnancy seizures are likely
to experience epilepsy-related adverse effects
including status epilepticus and increased epileptic
seizures(2). Uncontrolled seizures influence
maternal and fetal mortality and morbidity;
tonic-clonic seizures may cause physical damage
and spontaneous abortion , hypoxia, acidosis
and intracranial hemorrhage in the fetus; also
fetal bradycardia is possible during the mother’s
seizures. The etiology of epileptic seizures
in pregnancy includes epilepsy of unknown cause,
metabolic disorders, eclampsia, and cerebral
sinus thrombosis, causing pregnancy and postpartum
seizures(4, 6). Convulsion has occurred in 0.5-1%
of pregnancies(6), and is one of the serious
neurologic disorders in pregnancy(6, 7).
The most seizure attacks occurred in pregnant
women, who have history of epilepsy (8). Another
study reported that 1 in 200 pregnant women
experience seizures(9).
In India, 2.5 million women are experiencing
epileptic seizures, almost half of whom are
at fertility ages(10). Approximately 1 million
women of childbearing age in the US have seizures,
among whom 20 thousand go into labor, annually
(2, 3, 5, 11, 12).
In a study in the US on 45,000 pregnant women,
21.4 cases per 1,000 experienced non-eclamptic
seizures before or during pregnancy(13). According
to estimations, 3 to 5 per 1,000 births are
related to women with epilepsy (2-4, 11) .
Another study in Europe in 2013 reported that
pregnant women comprise 25% of all epileptic
patients, and most of these women are in need
of long-term treatment with antiepileptic medications.
Approximately 3-4 out of 1,000 pregnancies concern
women with a history of epilepsy and 1,800-2,400
infants in Britain are born from women with
a history of epilepsy, most of whom have healthy
pregnancies and infants(14).
In a study conducted between 1991 and 2000,
at Vali-Asr Hospital of Tehran, out of 21,000
admitted pregnant women, 53 cases had epilepsy;
of these women 55% were nulliparous and 45%
multiparous; 82% had generalized epilepsy, 14%
had focal epilepsy, and 4% had other types.
Epileptic attacks occurred in 34 subjects (70%)
during pregnancy, and the number of attacks
increased in only 15 patients (32%) and declined
in 9.5%; 58.6% showed no change in the seizure
frequency. This study reported that the cause
of increased epileptic attacks may be due to
the discontinuation of anti-epileptic medication
and pharmacokinetic and pharmacodynamic changes
of these medications during pregnancy, as well
as, insomnia, stress, and anxiety(10) .
Epilepsy control must be handled adequately,
as the frequency of attacks increases in 15
to 30% of the cases. Pregnancy-induced changes
of anti-epileptic medications are an important
agent(8). Due to reduced serum albumin and increased
hepatic and renal clearance, and increased emission
volume, the blood level of anti-epileptic drugs
falls in pregnancy(5, 8, 9, 12, 15), causing
a changed control over epileptic attacks in
pregnancy. Mother’s compliance and acceptance
is, however, another important factor (5, 15).
Fear of fetal adverse effects is a major issue
in pregnancy(12), accompanied by nausea, vomiting,
and sleep disorders of the mother(5). One of
the most important predicting factors is the
frequency of attacks in pregnancy versus the
prepartum years(16).
It has been shown that sex hormones influence
epileptic attacks, as estrogen decreases the
threshold and progesterone increases it (17).
Nearly 1-2% of epileptic women are afflicted
with status epilepticus, which causes mortality
and morbidity (8).
Preconception counseling, as well as monitoring
drug serum levels, along with drug and dosage
adjustment and providing patients with the information
concerning their condition, can help decrease
the frequency of attacks (10).
Seizures are the most probable to appear in
the first trimester of pregnancy and upon delivery
(8). Women with epilepsy who take anti-epileptic
medication are likely to suffer from an increased
risk of caesarean and hemorrhage(12, 15), yet
they do not experience increased risks of premature
contractions or preterm labor (2).
The present study was conducted in Kerman with
regard to the prevalence of pregnancy seizures
and the importance of maternal and fetal adverse
effects. It must be mentioned that this study
is unprecedented.
Patient Selection: This is a descriptive-analytical
study. The participants of the study were women
who referred to delivery centers of Kerman (public
and private) for childbirth. The patients were
initially asked to fill out the questionnaire
developed by Placencia et al (18-20) for screening,
containing 9 questions. The questionnaires were
also filled out by Hospital obstetricians, who
had previously received the required instructions
by a faculty member neurologist. Suspicious
patients were then examined by a neurologist
who filled out the complementary questionnaire,
including the demographic information of the
patients, type of epilepsy, history of attacks,
pregnancy condition, and medications used. A
case of epilepsy was approved by biography,
physical examination, and EEG. Patients with
approved epilepsy underwent lab assessment,
imaging, including MRI and CT scan, and lumbar
puncture, when required.
Sample Size: Considering the fact that
epileptic seizures are reported in 1% of all
pregnancies (5), the population of this study
comprised 3,800 cases admitted to public and
private hospitals. Since the cause of epileptic
seizures in pregnant women is different from
others and certain types are more frequent in
pregnancy, there was no need for a control group
in this study.
Exclusion Criteria: The exclusion criteria
were non-cooperation and dissent of participants
to sit interviews or non-cooperation of the
pertaining delivery wards for patient screening.
This study aimed at determining different types
of epilepsy (idiopathic or secondary causes)
and comparing epilepsy prevalence in nulliparous
and multiparous cases, as well as between pregnant
women with a prepartum history of seizures and
those with no such history. Furthermore, the
comparison of epilepsy prevalence according
to the admitted medical centers (public and
private hospitals) was determined according
to the prevalence of seizures in pregnant women
in each subgroup and a 95% confidence interval.
Comparison of prevalence between the groups
was carried out using chi-squared test.
This study was conducted subsequent to obtaining
informed consent from the participants, describing
the study objectives, and commitment to confidentiality.
Out
of
3,807
pregnant
women
who
were
admitted
to
public
and
private
hospitals
of
Kerman
in
the
last
month
of
their
pregnancy,
38
subjects
had
experienced
seizures
(epileptic
patients
and
those
who
experienced
their
first
seizure
in
pregnancy).
Age
of
the
epileptic
patients
ranged
from
15
to
33
years,
at
an
average
age
of
28.1274
±
5.60687
years;
it
was
24.6579
±
6.14267
for
patients
with
epileptic
attacks,
and
28.1624
±
5.60687
for
patients
without
epileptic
attacks,
and
the
difference
was
significant
(P=0.001).
In
this
study,
out
of
the
epileptic
patients
(a
total
of
24),
11
cases
were
nulliparous
and
13
cases
were
multiparous,
while,
a
total
of
901
subjects
were
nulliparous
which
was
statistically
significant
(P=0.017).
A
number
of
2,125
cases
were
admitted
to
public
hospitals
and
1682
cases
referred
to
private
hospitals.
Incidence
of
epileptic
attacks
was
31
cases
among
those
admitted
to
public
hospitals
and
7
cases
in
those
admitted
to
private
ones,
indicating
a
significant
difference
(P=0.001).
(Table
1).
Table
1:
Relationship
between
education
and
seizures
in
pregnant
women
and
type
of
Hospital
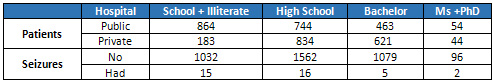
The
degree
of
gravidity
and
parity
in
customers
of
public
and
private
hospitals
showed
no
significant
difference,
yet,
the
number
of
abortions
was
higher
in
public
hospitals,
which
was
statistically
significant
(P=0.000)
(Table
2).
In
total
pregnant
women
these
factors
are
shown
in
Table
3.
Click
here
for
Table
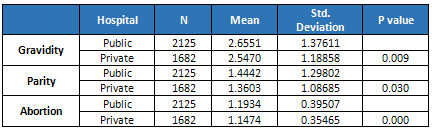
2:
Relationship
between
seizures
and
some
pregnancy
factors
Pregnant
women
with
higher
levels
of
education
referred
to
private
hospitals
more
(P=0.000).
The
pregnant
women
with
higher
education
have
lower
number
of
seizures
(P=0.039).
Table
3:
Relationship
between
hospital
type
and
a
number
of
pregnancy
factors
Customers
of
public
hospitals
had
lower
education.
The
majority
of
pregnant
women
in
the
public
sector
were
less
than
high
school
diploma,
while,
those
in
the
private
sector
held
higher
than
high
school
diploma
(P=0.000).
In
this
study,
the
number
of
epileptic
patients
was
directly
associated
with
the
education
level
(P=0.039)
(Table
1).
Out
of
the
24
epileptic
pregnant
women,
20
were
receiving
antiepileptic
medications
(7
patients
under
multi-drug
and
13
under
single-drug
regimens);
in
2
cases,
the
epilepsy
was
controlled
and
the
treatment
was
discontinued,
and
2
cases
did
not
take
any
medications.
Epileptic
attacks
were
controlled
in
only
1
case
among
the
multi-drug
patients
(%14),
while
they
were
controlled
in
7
cases
of
single-drug
patients
(54%),
approximately
4
times
the
former
group.
The
frequency
of
attacks
declined
in
5
cases
(21%),
increased
in
5
(21%),
and
was
unchanged
in
14
cases
(58%).
Of
all
the
patients
experiencing
epileptic
attacks,
24
had
epilepsy
(2
cases
did
not
take
antiepileptic
medications,
and
2
cases
had
discontinued
their
medications
after
controlling
seizures),
13
cases
were
non-epileptic,
and
1
case
had
a
childhood
experience
of
febrile
seizure.
Among
the
38
cases
with
a
history
of
seizures,
27
cases
had
seizures
in
pregnancy,
and
the
remaining
11
cases
experienced
no
seizures
in
pregnancy;
6
cases
had
a
family
history
of
epilepsy.
Out
of
the
27
women
with
pregnancy
seizures,
13
subjects
had
a
history
of
epilepsy,
14
cases
experienced
seizures
for
the
first
time
in
their
pregnancy;
out
of
11
cases
without
pregnancy
seizures,
8
cases
had
a
history
of
active
epilepsy.
In
2
cases
who
did
not
take
medications
seizures
were
controlled,
and
one
subject
had
an
experience
of
seizures
once
in
her
childhood.
Out
of
22
patients
with
active
epilepsy,
5
had
a
history
of
myoclonic
jerk.
No
myoclonic
jerks
were
reported
in
patients
with
inactive
epilepsy
or
those
experiencing
their
first
epileptic
seizure
in
pregnancy.
Table
4
portrays
the
occurrence
of
different
epileptic
seizures
in
the
patients.
Click
here
for
Table
4:
Type
of
seizures
in
pregnant
women
Concerning
the
anti-epileptic
medications,
13
cases
received
single-drug
regimens
(9
cases,
carbamazepine;
2
cases,
lamotrigine;
2
cases
sodium
valproate)
and
7
subjects
received
multi-drug
regimens
(3
cases,
lamotrigine
and
levetiracetam;
2
cases,
carbamazepine
and
levetiracetam;
1
case,
carbamazepine
and
sodium
valproate;
and
1
case,
carbamazepine
and
lamotrigine).
In
the
present
study,
11
cases
had
a
history
of
abortion,
out
of
whom:
2
cases
experienced
cerebral
vein
thrombosis
(CVT)
and
1
case,
eclampsia;
6
cases
had
epilepsy;
1
case
had
Arterio-Venous
Malformation
(AVM),
and
1
case
had
brain
tumor.
Six
cases
of
epileptic
patients
(24%)
had
a
history
of
abortion,
whereas,
in
the
remaining
participants,
654
cases
out
of
the
total
3783
subjects
(17%)
had
experienced
abortion
previously,
indicating
a
significant
difference
(P=0.000).
However,
there
was
no
significant
difference
in
the
increasing
cases
of
epilepsy,
between
pregnant
women
with
a
history
of
abortion
and
those
without
such
history
(P=0.111).
The
difference
between
the
degree
of
parity
and
gravidity
between
women
with
a
history
of
seizures
and
those
with
no
such
history
was
significant
(P=0.000)
(Table
2).
For
all
the
patients
with
a
history
of
eclampsia,
epilepsy,
CVT,
and
AVM,
pregnancy
seizures
occurred
in
the
third
trimester
(except
for
a
CVT
patient
who
experienced
seizures
in
month
2).
Only
5
out
of
38
patients
(total
number
of
patients)
had
an
abnormal
neurological
examination.
According
to
the
assessments,
one
case
was
caused
by
AVM,
another
case
by
trauma,
one
case
by
tumor;
epilepsy
in
24
cases
had
unknown
etiology.
The
causes
of
the
first
seizures
in
pregnancy
were
eclampsia
in
7
cases,
CVT
in
3
cases,
and
TTP
in
one
case.
In
the
present
study,
out
of
3807
pregnant
women
admitted
to
public
and
private
hospitals
of
Kerman
in
their
last
month
of
pregnancy,
38
cases
(0.99%)
had
previously
experienced
seizures.
In
a
study
in
the
US
on
45,000
pregnant
women,
21.4
cases
per
1,000
individuals
were
reported
to
have
experienced
one
non-eclamptic
seizure
during
or
before
pregnancy(2).
A
study
in
Vali
Asr
Hospital
of
Tehran
in
1991,
reported
53
epileptic
cases
out
of
21,000
pregnant
women
(10).
The
pregnancy
prevalence
in
a
study
in
London
was
reported
at
0.5-1%(21).
An
Australian
study
claimed
that
1
in
200
pregnant
women
experience
seizures(9).
Out
of
the
total
referrals,
2,125
women
were
admitted
to
public
hospitals
and
1,682
women
to
private
hospitals.
The
prevalence
of
seizures
was
significantly
higher
in
patients
admitted
to
public
hospitals
(P=0.001).
Numerous
causes
to
this
are:
1.
Patients
with
complications
who
are
more
likely
to
experience
epilepsy
are
referred
to
teaching
(public)
hospitals;
2.
Patients
of
private
hospitals
hold
higher
education,
and
according
to
studies
in
this
region,
lower
education
is
directly
associated
with
epilepsy;
3.
This
study,
also,
revealed
that
the
education
of
pregnant
women
in
public
hospitals
is
lower,
matching
the
results
of
a
2011
study
in
Kerman,
indicating
a
significant
relationship
between
the
incidence
of
epilepsy
and
lower
education
(22).
Our
findings
were
similar
to
those
of
studies
conducted
in
the
US
(23),
Vietnam(24),
and
Turkey
(25),
whereas,
in
his
study
in
England,
Ferro
did
not
report
such
a
relationship
(26);
4.
The
degrees
of
gravidity
and
parity
and
abortion
were
higher
in
women
admitted
to
public
hospitals,
which
was
only
significant
in
case
of
abortion.
This
may
have
caused
the
increased
cases
of
epilepsy.
In
a
study
in
Canada
on
55
pregnant
epileptic
women,
it
was
shown
that
42
patients
were
admitted
to
public
hospitals
and
13
patients
to
private
hospitals
(P=0.000)(27).
The
age
of
our
patients
ranged
from
15
to
33.
A
study
in
Italy
reported
the
age
of
pregnant
epileptic
patients
between
15.3
to
43.5
years
(28).
The
decline
of
the
childbearing
age
in
Iran
is
due
to
cultural
conditions.
The
important
point
is
that,
the
age
of
pregnant
women
who
experienced
epileptic
seizures
was
lower
than
others
(24.6
years
compared
to
28.1
years).
A
study
in
England
reported
the
average
age
of
epileptic
patients
(38
cases)
at
between
11
to
35
years
old(29).
The
average
age
of
pregnant
epileptic
patients
is
reported
at
26
years
by
a
study
in
India
(30).
This
difference
arises
from
cultural
conditions.
Among
our
patients,
6
cases
(15.7%)
had
a
family
history
of
epilepsy.
This
rate
was
17.3%
in
a
study
in
Kerman,
in
2011
(22),
close
to
that
of
a
study
in
Turkey
(14.3%)
(25).
In
the
present
study,
out
of
patients
with
a
history
of
epilepsy
(24
cases),
14
patients
were
multiparous
and
10
cases
were
nulliparous,
while,
901
women
in
the
population
were
nulliparous,
indicating
a
significant
difference.
A
study
in
England
reported
12
multiparous
cases
out
of
38
participants
(P=0.000)
(29).
In
another
study
in
Canada
it
was
observed
that
there
was
no
relationship
between
worsening
of
seizures
and
the
childbearing
age,
parity,
toxemia,
a
family
history
of
epilepsy,
age
at
onset
of
pregnancy,
and
preterm
labor.
The
most
prevalent
complication
in
these
patients
was
preterm
labor,
occurring
in
8
cases
(27).
Preterm
labor
or
abortion
occurred
in
2.6%
of
the
cases
in
a
study
in
Italy
(28).
Eleven
of
our
cases
had
a
history
of
abortion,
out
of
whom:
2
cases
experienced
cerebral
vein
thrombosis
(CVT)
and
1
case,
eclampsia;
6
cases
had
epilepsy;
1
case
with
Arterio-Venous
Malformation
(AVM),
and
1
case
had
brain
tumor.
Six
cases
of
the
epileptic
patients
(24%)
had
a
history
of
abortion,
while,
for
the
remaining
participants,
654
cases,
out
of
the
total
3,783
subjects
(17%),
had
previously
experienced
abortion,
indicating
a
significant
difference
(P=0.000).
However,
there
was
no
significant
difference
in
the
increasing
cases
of
epilepsy,
between
pregnant
women
with
a
history
of
abortion
and
those
without
such
history
(P=0.111).
We
observed
a
significant
difference
between
the
degree
of
parity
and
gravidity
between
women
with
and
without
a
history
of
seizures
(P=0.000).
None
of
our
patients
experienced
status
epilepticus,
similar
to
our
Canadian
counterpart(27),
however,
in
the
study
conducted
in
Italy,
21
in
3,415
cases
(0.6%)
experienced
status
epilepticus
(28).
The
similar
study
in
England
reported
2
patients
with
status
epilepticus
(29).
For
all
the
patients
with
a
history
of
eclampsia,
epilepsy,
CVT,
and
AVM,
pregnancy
seizures
occurred
in
the
third
trimester
(except
for
a
CVT
patient
who
experienced
seizures
in
month
2).
The
study
in
Canada
reported
exacerbation
of
seizures
in
early
pregnancy
(27).
In
England,
the
frequency
of
seizures
rose
in
the
first
trimester
(29).
Yet,
in
Italy,
the
exacerbation
of
seizures
was
reported
in
29%
of
the
cases
in
the
first
trimester,
32%
in
the
second
trimester,
and
39%
in
the
third
trimester
(28).
We
have
no
explanations
for
these
differences.
Seizure
recurrence
declined
in
5
cases
(20%),
increased
in
5
cases
(20%),
and
remained
constant
in
15
cases
60%
of
the
subjects.
In
the
study
in
England,
out
of
38
pregnant
women
with
idiopathic
epilepsy,
seizure
frequency
increased
in
45.2%
of
the
cases,
remained
constant
in
50%,
and
declined
in
4.8%
(29).
Pregnancy
in
the
Australian
study
increased
seizures
in
24%
of
the
subjects(9).
Seizure
frequency
in
the
Canadian
study
increased
in
9,
remained
constant
in
14,
declined
in
4
cases,
and
was
unknown
for
the
rest
of
the
participants(27).
In
Italy,
pregnancy
seizures
were
unchanged
in
70.5%
of
the
patients,
declined
in
12%,
and
increased
in
15.8%
of
the
cases(28).
A
study
in
Texas
reported
a
decline
in
the
seizures
in
3-24%
of
the
subjects,
a
rise
in
14-32%,
and
no
change
in
54-80%;
84-92%
of
the
patients
were
pregnancy
seizure-free
(4).
The
study
in
England
showed
a
positive
relationship
between
seizure
frequency
2
years
prior
to
pregnancy
and
increased
seizure
frequency
in
pregnancy.
Patients
with
more
than
one
seizure
per
month
had
a
higher
risk
of
exacerbation
in
pregnancy,
whereas,
only
25%
of
patients
whose
seizure
intervals
were
more
than
9
months,
got
worse
(29).
However,
in
the
study
by
Rosciszewka
and
Grudzinska
in
1970,
no
such
relationship
was
reported
(21).The
average
age
of
onset
of
seizures
was
lower
in
subjects
with
increased
seizures
(12.6
years
old),
compared
to
others
(16.1
years),
yet,
insignificant.
In
the
present
study
we
did
not
take
into
account
the
age
of
onset
of
seizures
in
epileptic
patients.
Of
the
referrals,
24
cases
were
epileptic,
with
2
cases
of
inactive
and
22
cases
of
active
epilepsy.
Out
of
the
total
patients,
25
cases
had
a
history
of
seizures,
while
13
cases
did
not.
Out
of
the
total
27
cases
with
pregnancy
epilepsy,
13
cases
already
had
epilepsy,
while,
14
cases
experienced
seizures
for
the
first
time
in
their
pregnancy.
From
the
38
cases
with
previous
seizures,
11
cases
had
no
pregnancy
seizures
(8
cases
had
a
history
of
active
epilepsy,
2
cases
had
non
active
epilepsy,
and
one
of
the
patients
experienced
epilepsy
in
her
childhood
only
once),
while,
27
cases
experienced
pregnancy
seizures.
Six
cases
had
a
family
history
of
epilepsy
and
32
had
no
such
history.
In
a
study
in
England,
in
1974,
59
pregnant
women
had
epilepsy,
out
of
whom,
14
cases
experienced
their
first
seizure
in
pregnancy:
7
cases
in
the
first
pregnancy,
5
cases
in
the
second,
one
case
in
the
third,
and
one
case
in
the
fifth
pregnancy.
Thirty
eight
cases
had
idiopathic
and
7
cases
had
symptomatic
epilepsy.
In
the
former
group,
11
cases
had
only
one
seizure,
and
5
cases
had
recurrent
seizures
in
the
very
same
pregnancy
(29).
In
Canada,
55
patients
had
a
history
of
idiopathic
epilepsy
and
3
cases
experienced
seizures
in
pregnancy
for
the
first
time
(27).
In
our
study
seizure
attacks
occurred
in
the
third
trimester
of
pregnancy,
except
one
case
of
CVT,
which
was
in
the
second
month.
The
8
cases
who
were
epileptic
had
frequent
attacks.
The
England
study
reported
pregnancy
seizures
from
week
10
to
38,
one
case
upon
delivery,
and
one
case
postpartum
(29).
None
of
the
cases
had
seizures
upon
delivery
in
the
study
in
Australia
and
seizure
recurrence
in
pregnancy
was
reported
between
30
and
50%
in
the
study
conducted
in
Australia
(9).
Incidence
of
different
seizures
is
portrayed
in
Table
4
(76.3%
GTC
(generalized
tonic-clonic),
13.2%
Juvenile
Myoclonic
Epilepsy
(JME),
and
2.6%
Focal-Generalized,
and
7.9%
Complex
Partial
Seizures
(CPS).
In
the
22
patients
with
active
epilepsy,
5
cases
had
myoclonic
jerks,
and
17
cases
did
not.
No
myoclonic
jerks
was
reported
in
patients
with
inactive
epilepsy
or
those
experiencing
their
first
epileptic
seizure
in
pregnancy.
In
Australia,
in
a
study
on
75
pregnant
epileptic
women,
the
majority
of
the
cases
(82.7%)
had
GTC
(9).
The
study
in
Canada
reported
most
patients
with
GTC;
two
cases
had
absence,
and
4
cases
had
focal
or
psychomotor
epilepsy
(27).
In
Italy,
GTC
was
observed
in
39.3%
of
the
cases,
localized
in
47.1%,
and
unknown
in
the
rest
(28).
Etiology
of
Epilepsy
in
the
Present
Study:
Only
5
out
of
our
total
38
patients
had
abnormal
neurological
examination.
According
to
the
assessments,
one
case
was
caused
by
AVM,
another
case
by
head
trauma,
one
case
by
tumor;
epilepsy
in
24
cases
had
unknown
etiology.
The
causes
of
the
first
seizures
in
pregnancy
were
eclampsia
in
7
cases,
CVT
in
3
cases,
and
TTP
in
one
case.
The
English
study
reported
the
etiology
of
symptomatic
epilepsy
as
including
meningitis,
brain
abscess,
encephalitis,
meningioma,
and
head
trauma,
none
of
which
was
observed
in
the
present
study
(29).
Epilepsy
in
the
study
in
Canada
was
caused
by
head
trauma
in
5
cases,
and
brain
aneurysm
in
4
cases
(27).
In
a
study
done
in
Africa,
prevalence
of
eclampsia
was
reported
at
1.02%
(31).
In
this
study,
epileptic
patients
mostly
used
Carbamazepine,
in
addition
to
a
few
cases
of
Lamotrigine,
Levetiracetam,
and
Sodium
Valproate.
Seven
cases
received
multi-drug
regimens
(2
drugs),
and
13
cases
received
single-drug
regimens.
Seizures
were
seen
in
1
case
of
single-drug
patients
(14%),
and
7
cases
of
multiple-drug
cases
(54%),
almost
four
times
the
single-drug
patients.
In
Australia,
70.7%
of
pregnant
women
with
a
history
of
epilepsy
received
anti-epileptic
medications
prior
to
pregnancy.
Epilepsy
was
controlled
in
46
patients
with
only
one
or
two
drugs.
The
most
common
anti-epileptic
medication
was
Phenytoin
(81%),
followed
by
Phenobarbital
29.3%,
Primidone
20.7%,
Carbamazepine
13.8%,
and
Sodium
Valproate
5.2%.
Out
of
the
said
patients,
39.6%
received
only
one
medication,
39.6%
two
medications,
17.4%
three,
and
3.4%
more
than
three
medications
(9).
In
Italy,
recurrence
of
seizures,
mostly
GTC
seizures,
was
higher
in
those
receiving
Lamotrigine
(58.2%),
and
there
was
no
seizure
recurrence
in
75%
of
Valproate
users,
67.3%
of
Carbamazepine
users,
and
73.4%
of
Phenobarbital
users
(28).
More
than
6
in
1,000
pregnant
women
have
epilepsy.
Though
without
a
history
of
epilepsy,
less
than
4
in
1,000
pregnant
women
are
afflicted
with
seizures,
mostly
caused
by
eclampsia.
Seizures
increased
in
21%
of
epileptic
pregnant
patients,
and
declined
in
21%
of
the
cases.
1.
Rowland
P,
Pedley
A.
Merritt’s
neurology.
2010:927-41.
2.
Harden
C,
Hopp
J,
Ting
T,
Pennell
P,
French
J,
Hauser
WA,
et
al.
Practice
Parameter
update:
Management
issues
for
women
with
epilepsy—Focus
on
pregnancy
(an
evidence-based
review):
Obstetrical
complications
and
change
in
seizure
frequency
Report
of
the
Quality
Standards
Subcommittee
and
Therapeutics
and
Technology
Assessment
Subcommittee
of
the
American
Academy
of
Neurology
and
American
Epilepsy
Society.
Neurology.
2009;73(2):126-32.
3.
Harden
C,
Hopp
J,
Ting
T,
Pennell
P,
French
J,
Hauser
WA,
et
al.
Practice
Parameter
update:
Management
issues
for
women
with
epilepsy—Focus
on
pregnancy
(an
evidence-based
review):
Obstetrical
complications
and
change
in
seizure
frequency.
Report
of
the
Quality
Standards
Subcommittee
and
Therapeutics
and
Technology
Assessment
Subcommittee
of
the
American
Academy
of
Neurology
and
American
Epilepsy
Society.
Neurology.
2009;73(2):133-41.
4.
Hart
LA,
Sibai
BM,
editors.
Seizures
in
pregnancy:
epilepsy,
eclampsia,
and
stroke.
Seminars
in
perinatology;
2013:
Elsevier.
5.
Pschirrer
ER,
Monga
M.
Seizure
disorders
in
pregnancy.
Obstetrics
and
gynecology
clinics
of
North
America.
2001;28(3):601-11.
6.
Pandey
R,
Garg
R,
Darlong
DV,
Punj
J,
Khanna
P.
Recurrent
Seizures
in
Pregnancy—Epilepsy
or
Eclampsia:
A
Diagnostic
Dilemma?
AANA
journal.
2011;79(5):389.
7.
Walker
S,
Permezel
M,
Berkovic
S.
The
management
of
epilepsy
in
pregnancy.
BJOG:
An
International
Journal
of
Obstetrics
&
Gynaecology.
2009;116(6):758-67.
8.
Beach
RL,
Kaplan
PW.
Seizures
in
pregnancy:
diagnosis
and
management.
International
review
of
neurobiology.
2008;83:259-71.
9.
Svigos
J.
Epilepsy
and
pregnancy.
Australian
and
New
Zealand
Journal
of
Obstetrics
and
Gynaecology.
1984;24(3):182-5.
10.
Borna
S,
Khazardoost
S,
Hantoushzadeh
S,
Borna
H.
The
course
and
outcome
of
pregnancy
in
women
with
epilepsy
in
Valie-Asr
Hospital.
Iranian
Red
Crescent
Medical
Journal.
2006;8:36-40.
11.
Kamyar
M,
Varner
M.
Epilepsy
in
pregnancy.
Clinical
obstetrics
and
gynecology.
2013;56(2):330-41.
12.
Caughey
AB.
Seizure
disorders
in
pregnancy.
E
Medicine.
2013.
13.
Nelson
KB,
Ellenberg
JH.
Maternal
seizure
disorder,
outcome
of
pregnancy,
and
neurologic
abnormalities
in
the
children.
Neurology.
1982;32(11):1247-.
14.
Dondapati
S,
Baban
M,
Haque
S.
A
descriptive
case
of
seizure
in
pregnancy:11AP3-5.
European
Journal
of
Anaesthesiology
(EJA
).
2013:30:172-3.
15.
Dalessio
DJ.
Seizure
disorders
and
pregnancy.
New
England
Journal
of
Medicine.
1985;312(9):559-63.
16.
Bui
E,
Klein
A.
women
with
epilepsy:
a
practical
management
Handbook:
Cambridge
University
Press.
2014.
17.
Morrell
MJ.
Epilepsy
in
women.
American
family
physician.
2002;66(8):1489-94.
18.
Placencia
M,
Sander
J,
Shorvon
S,
Ellison
R,
Cascante
S.
Validation
of
a
screening
questionnaire
for
the
detection
of
epileptic
seizures
in
epidemiological
studies.
Brain.
1992;115(3):783-94.
19.
Placencia
M,
Sander
J,
Shorvon
S,
Ellison
R,
Cascante
S.
Validation
of
a
screening
questionnaire
for
the
detection
of
epileptic
seizures
in
epidemiological
studies.
Brain.
1992;115(3):771-82.
20.
Placencia
M,
Suarez
J,
Crespo
F,
Sander
J,
Shorvon
S,
Ellison
R,
et
al.
A
large-scale
study
of
epilepsy
in
Ecuador:
methodological
aspects.
Neuroepidemiology.
1992;11(2):74-84.
21.
Rosciszewska
D.
[Epilepsy
and
pregnancy].
Polski
tygodnik
lekarski
(Warsaw,
Poland
:
1960).
1994;49(4-5):102-3.
22.
Ebrahimi
H,
Shafa
M,
Hakimzadeh
Asl
S.
Prevalence
of
active
epilepsy
in
Kerman,
Iran:
a
house
based
survey.
Acta
Neurol
Taiwan.
2012;21(3):115-24.
23.
Kobau
R,
Zahran
H,
Thurman
DJ,
Zack
MM,
Henry
TR,
Schachter
SC,
et
al.
Epilepsy
surveillance
among
adults--19
States,
behavioral
risk
factor
surveillance
system,
2005:
US
Department
of
Health
and
Human
Services,
Centers
for
Disease
Control
and
Prevention;
2008.
24.
LE
QC,
Nguyen
VH,
Jallon
P.
Prevalence
of
epilepsy
in
Phu
Linh-Soc
Son–Hanoi,
a
rural
region
in
North
Vietnam.
Neurology
Asia.
2007;12(Supplement
1):57.
25.
Velioglu
SK,
Bakirdemir
M,
Can
G,
Topbas
M.
Prevalence
of
epilepsy
in
northeast
Turkey.
Epileptic
disorders.
2010;12(1):22-37.
26.
Ferro
MA.
A
population-based
study
of
the
prevalence
and
sociodemographic
risk
factors
of
self-reported
epilepsy
among
adults
in
the
United
Kingdom.
Seizure.
2011;20(10):784-8.
27.
Sabin
M,
Oxorn
H.
Epilepsy
and
pregnancy.
Obstetrics
&
Gynecology.
1956;7(2):175-9.
28.
Battino
D,
Tomson
T,
Bonizzoni
E,
Craig
J,
Lindhout
D,
Sabers
A,
et
al.
Seizure
control
and
treatment
changes
in
pregnancy:
observations
from
the
EURAP
epilepsy
pregnancy
registry.
Epilepsia.
2013;54(9):1621-7.
29.
Knight
A,
Rhind
E.
Epilepsy
and
pregnancy:
a
study
of
153
pregnancies
in
59
patients.
Epilepsia.
1975;16(1):99-110.
30.
Thomas
S,
Indrani
L,
Devi
G,
Jacob
S,
Beegum
J,
Jacob
P,
et
al.
Pregnancy
in
women
with
epilepsy:
preliminary
results
of
Kerala
registry
of
epilepsy
and
pregnancy.
Neurology
India.
2001;49(1):60-6.
31.
Tukur
J,
Muhammad
Z.
Management
of
eclampsia
at
AKTH:
before
and
after
magnesium
sulphate.
Nigerian
Journal
of
Medicine.
2010;19(1).
|

